Auroras are not just beautiful light displays—they are electric, chemical reactions happening high above Earth. Powered by solar storms and guided by Earth’s magnetic field, different gases glow in distinct colors as energized atoms release light. This in-depth guide explains the shocking chemistry behind aurora colors, why some hues are rare, and what these cosmic colors reveal about space weather.
When the Night Sky Becomes a Living Work of Art
There are few moments in life when people stop everything they’re doing, look up, and fall completely silent. The aurora does that.
Whether seen from Alaska, Minnesota, Iceland, or unexpectedly from much farther south, auroras don’t just decorate the sky—they transform it. Curtains of green ripple like silk. Red glows simmer quietly near the horizon. Purple flashes spark and vanish in seconds.
It feels artistic. Emotional. Almost intentional.
Yet auroras are not painted by hand or guided by intelligence. They are created by electricity and chemistry, acting together on a planetary scale. Every color you see is the result of an atomic interaction—electrons jumping, energy releasing, and gases glowing in precise ways.
This article explains the real science behind aurora colors, why green dominates, why red is rare and unsettling, why purple appears during intense storms, and why scientists consider auroras one of the most honest expressions of physics in the universe.
What an Aurora Really Is (Beyond the Simplified Explanation)
Most explanations reduce auroras to a single sentence: charged particles from the Sun hit Earth’s atmosphere and create light.
That statement is true—but dangerously incomplete.
An aurora is actually the final step in a long chain of events involving:
- Solar storms releasing massive energy
- Charged particles traveling through space
- Earth’s magnetic field funneling that energy
- Atmospheric atoms absorbing and releasing energy
- Human eyes and cameras interpreting color
In simple terms, auroras are electric reactions written in chemistry and displayed as color.
Where Aurora Energy Comes From: The Sun’s Invisible Power
Auroras don’t begin with light. They begin with magnetic energy on the Sun.
The Sun constantly releases charged particles through the solar wind. During periods of intense activity—solar flares and coronal mass ejections (CMEs)—this flow becomes explosive.
These particles:
- Carry electrical charge
- Travel at extreme speeds
- Are guided by magnetic fields
- Interact violently with Earth’s magnetosphere
When this energy reaches Earth, it doesn’t hit the atmosphere evenly. Earth’s magnetic field decides where it goes.
Earth’s Magnetic Field: The Curator of the Sky’s Art
Earth’s magnetic field acts like a cosmic funnel.
Instead of letting solar particles strike the atmosphere everywhere, it:
- Redirects them toward the poles
- Channels them along invisible magnetic lines
- Concentrates energy into auroral zones
That’s why auroras usually appear near the Arctic and Antarctic.
During strong solar storms, this funnel widens—allowing auroras to appear far south of their normal range.
The Electric Moment That Creates Light
When solar particles finally collide with Earth’s upper atmosphere, they crash into atoms and molecules at incredible speeds.
These collisions:
- Transfer energy to atmospheric atoms
- Knock electrons into higher energy states
- Leave atoms temporarily unstable
Atoms don’t like being unstable.
When electrons fall back to their normal positions, the excess energy is released as light.
That light—its color, brightness, and movement—is the aurora.
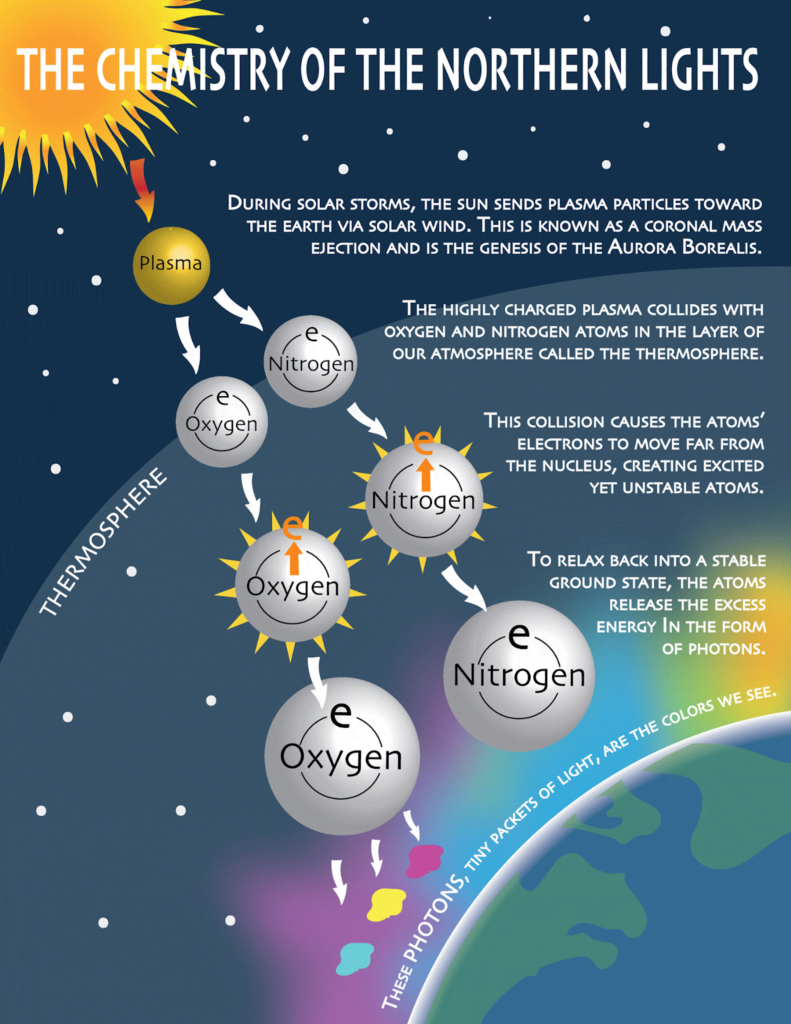
Why Aurora Colors Are Chemistry, Not Random Beauty
Every aurora color corresponds to a specific gas, altitude, and energy level.
Auroras are not painted in broad strokes. They are precise chemical reactions, happening millions of times per second.
Green Auroras: The Signature Color of the Northern Lights
Green is the most common aurora color—and the one most people recognize.
Green auroras are produced by:
- Oxygen atoms
- At altitudes of roughly 60–120 miles
- Moderate-energy particle collisions
When oxygen atoms at these altitudes are excited, they emit a greenish-yellow light as electrons return to normal energy levels.
Green dominates because:
- Oxygen is abundant
- The energy required is relatively easy to achieve
- Human eyes are especially sensitive to green in low light
This is why even faint auroras usually appear green.
Red Auroras: Rare, Quiet, and Unsettling
Red auroras feel different. They don’t dance as much. They glow.
Red auroras come from:
- Oxygen atoms
- At extremely high altitudes (150–300 miles)
- Very thin air
- Extremely high energy input
At these heights, oxygen atoms release energy more slowly, producing a deep red glow.
Because the air is so thin, red auroras require powerful solar storms to form. That’s why red auroras are rare—and why they often appear during historic space-weather events.
Purple, Blue, and Pink Auroras: Nitrogen Takes Over
Purple, blue, and pink colors usually appear at the edges or lower borders of auroras.
These colors are produced by:
- Nitrogen molecules
- Lower altitudes
- Very energetic particle collisions
Nitrogen emits:
- Blue and purple light when excited
- Pink tones when mixed with red oxygen above
These colors often appear during fast, violent auroral surges—when energy floods the atmosphere suddenly.
Why Yellow Auroras Are So Uncommon
Occasionally, auroras appear yellow or gold. This is not a separate chemical reaction.
Yellow auroras occur when:
- Green oxygen emissions
- Overlap with red emissions
- Blend visually in the observer’s eye
The result is a rare and striking hue—but it’s more optical mixing than pure chemistry.
Why Auroras Look Better in Photos Than in Person
Many people are surprised when their aurora photos look brighter than what they remember seeing.
This happens because of biology, not disappointment.
Human eyes:
- Lose color sensitivity in darkness
- Struggle with red light at night
- Prioritize motion over color
Cameras:
- Collect light over several seconds
- Amplify faint colors
- Reveal details invisible to the naked eye
That’s why some auroras feel subtle until you see them on a screen.
Why Auroras Move Like Living Creatures
Auroras don’t drift like clouds. They ripple, snap, pulse, and collapse.
This motion happens because auroras follow electric currents flowing along magnetic field lines.
When those currents fluctuate—even slightly—the aurora responds instantly.
What feels alive is actually electromagnetism in motion.
Real-Life Examples: When Aurora Colors Shocked the US
During recent geomagnetic storms, Americans reported:
- Crimson skies in Texas and Alabama
- Purple flashes over the Midwest
- Green curtains visible in California
Many thought they were seeing fires or city lights—until photos confirmed rare aurora colors.
These events demonstrated just how extreme the atmospheric chemistry had become.
Why Stronger Solar Storms Mean More Colors
Mild solar storms usually produce simple green auroras.
Stronger storms:
- Deliver higher-energy particles
- Penetrate deeper into the atmosphere
- Trigger multiple chemical reactions
That’s why intense auroras display red, purple, pink, and blue colors simultaneously.
What Aurora Colors Tell Scientists
Aurora colors are not just beautiful—they’re informative.
Scientists use them to:
- Measure storm strength
- Identify altitude of energy input
- Study atmospheric composition
- Track magnetic field behavior
Auroras are natural diagnostic tools for space weather.
Are Aurora Colors Dangerous?
Auroras are completely safe to observe.
The reactions occur far above:
- Aircraft
- Weather systems
- Human exposure
Earth’s atmosphere absorbs harmful radiation long before it reaches the ground.
Why Scientists Love Studying Aurora Chemistry
Auroras provide:
- A visible plasma laboratory
- A natural particle accelerator
- Real-time magnetic field visualization
They allow scientists to study fundamental physics without instruments—just observation.
10 Trending & Relevant FAQs About Aurora Colors
1. What causes different aurora colors?
Different gases and energy levels create different colors.
2. Why are auroras usually green?
Green oxygen emissions are easiest to produce and see.
3. What causes red auroras?
High-altitude oxygen excited by extreme energy.
4. Are purple auroras rare?
Yes, they require strong nitrogen excitation.
5. Can auroras appear blue or pink?
Yes, due to nitrogen and color mixing.
6. Why do auroras look brighter in photos?
Cameras collect more light than human eyes.
7. Do aurora colors indicate storm strength?
Yes—red and purple often signal stronger storms.
8. Are aurora colors the same in the Southern Hemisphere?
Yes, the chemistry is identical.
9. Can auroras change color rapidly?
Yes, as energy input fluctuates.
10. Are auroras safe to watch?
Yes, they pose no danger to people.
Why Auroras Are the Universe’s Most Honest Art
Auroras don’t hide their process.
Every color is:
- An atomic collision
- An electron releasing energy
- Physics made visible
No filters. No illusions. Just nature expressing energy as light.

Final Reflection: When Chemistry Becomes Cosmic Art
Auroras prove that science and beauty are not opposites.
Electricity becomes motion. Chemistry becomes color. Invisible forces become visible wonder.
When you watch an aurora, you are watching atoms glow because the Sun touched Earth.
That is not just beautiful.
That is unforgettable.




